Electron Microscopy - Verios 460 SEM

PPMS System Name: Verios 460 SEM
Department and Room: Physiology, Development and Neuroscience, Anatomy Building, Room 21
Voltages: Accelerating voltage range of 1-30 keV, plus low voltage range of 350 – 1000 eV
Emission source: Field-emission gun
Stage: Computer-driven, high precision stage with 360 degree continuous rotation and +60 degree to -10 degree stage tilt
Detectors/Cameras:
- Everhart-Thornley (ETD) and Through-Lens detectors (TLD) for routine SE- and BSE-imaging in field-free mode (large field of view/depth of field) and immersion-mode (high resolution imaging)
- Concentric backscatter detector (CBS) for differential detection of BS electrons scattered at various angles from the beam axis; custom-mode allows selection of individual detector segments
- Segmented STEM detector (STEM III) for TEM grid samples with custom settings for bright-field (BF), dark-field (DF) and high-angle annular dark field (HAADF) imaging
- Energy-dispersive X-ray detector (Ametek window-less silicon drift detector) for elemental analysis running with EDAX Genesis or TEAM software
- Mirror (MD) and In-column (ICD) detectors allowing detection of BS electrons for increasingly high Z-contrast imaging
Capabilities:
- Large range of image resolution and scan speed settings; custom settings for interlace scanning, multiple image integration and drift correction to improve image acquisition even of charging samples; definition of scan presets for easy and quick sample navigation and image acquisition
- UniColor mode (UC mode, monochromator, selectable below 5 keV/25 pA) to improve image quality at low keV/probe current settings
- Energy-dispersive X-ray detector (Ametek window-less silicon drift detector) for elemental analysis running with EDAX Genesis or TEAM software.
- Cryo-SEM: Quorum cryo-transfer system PP3010T for loading, coating and imaging quick-slush frozen, hydrated specimens
- Beam-deceleration mode in room temperature and cryo-mode
- MAPS software for automated acquisition of high resolution images from large areas of SEM specimens or TEM blockfaces
Back to main Electron Microscopy page
Secondary Electron Imaging
Backscatter Imaging
Imaging of Unstained Samples
Cryo-SEM
Scanning Transmissions Electron Microscopy Imaging
Blockface Imaging
Laser Ablation – SEM Blockface Imaging
SE/SEM-CLEM
Secondary Electron Imaging
Secondary electron imaging is the most frequently used mode in SEM to visualise specimen surfaces. Prior to imaging, samples are typically sputter-coated with conductive metals such as gold, platinum, iridium, or chromium. This coating reduces charging effects by enhancing electrical conductivity and improves image quality by increasing secondary electron yield, thus enhancing the signal-to-noise ratio. Additionally, the metal layer creates a uniform surface composition, making the secondary electron emission largely dependent on surface topography. This produces detailed, three-dimensional images with exceptional depth-of-field across a broad range of magnifications.
Drosophila intestinal tract (Golnar Kohlagar, Department of Physiology, Development and Neuroscience, Cambridge). The top overview image is a stitched map created using FEI MAPS software. Successive images show magnified structures denoted by the coloured squares.


Green square: longitudinal and circular muscle layers on the gut surface

Blue square: Mechanosensory bristle

Red square: ordered fibrillar structures near the crop
Backscatter Imaging
Backscatter electron (BSc) imaging is an alternative SEM modality that provides compositional (Z-contrast) information rather than solely surface topography. Elements with higher atomic numbers produce more backscattered electrons, appearing brighter in BSc images and highlighting elemental differences within samples. Typically, samples imaged in BSc mode can be left uncoated or coated with a thin carbon layer to enhance conductivity.
The Verios 460 SEM offers multiple backscatter detectors: the ETD and TLD detectors, which function in both SE and BSc modes; the concentric backscatter (CBS) detector, featuring segmented detection for angle-specific electron collection; and the dedicated mirror (MD) and in-column (ICD) detectors, positioned higher within the SEM column to generate increasingly pure compositional contrast images. BSc imaging is commonly paired with Energy-Dispersive X-ray (EDX) analysis to obtain comprehensive elemental information.

Carbon nanotubes imaged in SE-mode (left) showing their smooth surface topography. The image on the right is an overlay of this SE-image with the BSc image (red) revealing areas of high Z inside the CNTs. EDX showed this to be the iron catalyst used during CNT synthesis (Prof. Slawomir Boncel, Silesian University of Technology, Poland; DOI: 10.1021/acsbiomaterials.6b00197).

Partial fracture of freeze-dried wheat leaf. On the left, imaged in SE-mode using the ETD detector at 2 keV and 25 pA detailing mostly the surface topography of the sample. On the right, the leaf was imaged in BSc-mode using the CBS detector at 30 keV and 3.2 nA reaching more structures in the depth of the sample, e.g. intracellular chloroplasts (green square) or penetrating the surface wax layer (red square) to reveal the underlying epidermis cells (Karin Mueller, Microscopy Bioscience Platform-EM).
Imaging of Unstained Samples
For high-resolution imaging, such as nanoparticles or thin films, metal or carbon coatings become undesirable as they obscure fine surface details. However, imaging uncoated samples introduces challenges, notably charging effects in non-conductive specimens. Lowering accelerating voltages or probe currents can mitigate charging but reduces both signal-to-noise ratio and primary electron beam quality.
The Verios 460 addresses these issues with two specialized modes: UniColor (UC) and Beam Deceleration (BD). In UC mode, the electron beam passes through a monochromator, significantly reducing energy spread and minimizing chromatic aberration. BD mode applies a negative voltage bias to the specimen stage, decelerating electrons just before they reach the sample. This allows a higher accelerating voltage for improved beam quality while maintaining a lower electron landing energy, thus minimizing charging and beam damage. Additionally, the negative stage bias directs signal electrons towards detectors, enhancing signal-to-noise ratio.
Note that BD imaging performs best on flat samples, as rough surfaces can cause increased astigmatism and distortion. The Verios 460 supports a minimum accelerating voltage of 350 eV, minimum probe current of 0.8 pA, and stage bias adjustments between 50 and 4000 V. The Verios cryo-stage is also compatible with BD mode.

Octa-calcium phosphate imaged uncoated in UC/BD mode using a 2000 V stage bias (OCP-L-50%, Rui Li, Duer Group, Yusuf Hamied Dept. of Chemistry, Cambridge).
Cryo-SEM
The Verios 460 SEM includes a Quorum PP3010T cryo-preparation system, enabling imaging of soft, hydrated biological or material samples in their frozen state. A key advantage of cryo-SEM is minimal sample preparation without the need for prior fixation or dehydration; however, throughput is relatively low compared to other techniques. Samples are mounted on a cryo-SEM shuttle, plunge-frozen in slushed liquid nitrogen, and transferred to a cooled preparation stage. At this stage, specimens may be fractured, sublimed to remove surface ice, and sputter-coated (typically with platinum) to enhance conductivity and contrast. Finally, the prepared sample is moved to the SEM cryo-stage for imaging. Due to necessary run-up and run-down periods (~2 hours total, in addition to imaging), we recommend half-day or full-day bookings.

Examples of Cryo-SEM imaging:

Geranium petal, surface plus fracture at the petal base (Karin Müller, Microscopy Bioscience Platform-EM).
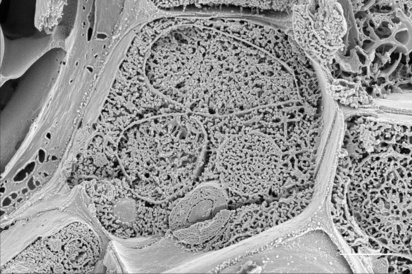
Plantain stem (fractured) showing cell walls and cell interior with various organelles and delineating membranes (Karin Müller, Microscopy Bioscience Platform-EM).

Curdled milk (fractured) showing round milk fat globules within a water/solute matrix (Karin Müller, Microscopy Bioscience Platform-EM).
Scanning Transmissions Electron Microscopy Imaging
The Verios 460 SEM features a retractable STEM detector designed for imaging thin, uncoated samples, particularly nanoparticles or dry powders, for Z-contrast analysis. The STEM insert accommodates up to six samples typically mounted on TEM holey or lacey carbon film grids. Its concentric detector design simultaneously captures bright-field, multiple dark-field, and high-angle annular dark-field (HAADF) images. The maximum accelerating voltage for STEM imaging is 30 keV.


Nanoparticles with a polydopamine shell and an empty core designed for drug delivery. Melanin is a natural pigment with diverse roles in various organisms. Polydopamine NPs are a synthetic mimic of naturally occurring melanin and are studied for tissue engineering, bioimaging, biosensing, catalysis and drug delivery applications. Pores can be introduced into this material by using templating agents significantly increasing its loading capacity (top: STEM BF and DF4 images, below: SE image of the same material in UC/BD mode). Andrea Bistrovic-Popov, Ljiljana Fruk Group, BioNano Engineering, Department of Chemical Engineering and Biotechnology, Cambridge
Blockface Imaging
SEM blockface imaging uses sample preparation protocols similar to those for TEM, but imaging is performed in a scanning electron microscope using backscatter mode. After fixation and heavy metal staining, samples are embedded in resin and mounted either as ultrathin sections on glass or plastic coverslips, or as intact resin blocks on SEM stubs. All specimens are sputter-coated with a thin carbon layer to reduce charging. Inverting the contrast of the resulting images produces visuals closely resembling TEM.
Compared to TEM, blockface imaging offers several advantages: it allows imaging of larger areas, avoids interference from grid bars, and provides a broader overview for locating regions of interest. However, the practical magnification is limited to ~8,000–10,000x. The Verios 460 SEM is equipped with FEI MAPS software for automated image tiling, enabling the generation of large, navigable specimen maps.

Map of a culture of green algae (Cosmarium subtumidum) revealing the morphology of the pyrenoids, the main actors of the algal carbon concentrating mechanism. Pyrenoids improve the efficiency of photosynthesis by delivering carbon dioxide to RuBisCo, an enzyme concentrated at the heart of this micro-compartment. Surrounded by a typical starch sheath, the pyrenoid is pervaded by an intricate network of microtubules (Myriam Goudet, Howard Griffiths, Physiological Ecology Group, Dept. of Plant Sciences, Cambridge).
Laser Ablation – SEM Blockface Imaging
During early embryonic development, elongation of the head-to-tail axis is a critical morphogenetic event. A study by Susannah McLaren (Steventon Lab, Department of Genetics, University of Cambridge; doi:10.1242/dev.199459) used light-mediated cell ablation alongside quantitative morphogenesis analysis to reveal that coordinated anterior notochord cell expansion and posterior progenitor addition generate forces driving axis elongation.
To assess the impact of ablation at high resolution, an unconventional correlative light and electron microscopy (CLEM) approach was used: laser-ablated regions were imaged using SEM blockface imaging. This revealed sharply defined zones of cellular damage, with precise fragmentation confined to the targeted area, highlighting the spatial accuracy of laser ablation.

Confocal image of a zebrafish embryo, the two ablations, a large area in the notochord and a small area in the tail bud, are highlighted in red. Following the ablation in a confocal laser microscope, the zebrafish embryos were processed for electron microscopy, sectioned and viewed by SEM blockface imaging with the aim to find the ablated areas.
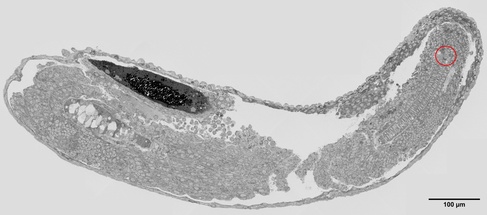
Low resolution map of a zebrafish embryo, the ablated area in the tail bud is circled in red. The larger ablation in the notochord, apparent in the confocal image, is not visible in this section as it lies more anteriorly along the axis at a different section depth. Once an ablated area is identified, the morphology of the tissue can be mapped in the SEM at increased magnifications.
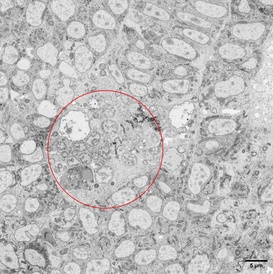
Medium resolution map of the zebrafish tail bud. The tissue area damaged by the laser is circled in red. The cell damage caused by the ablation becomes apparent at higher magnification. The close vicinity of normal versus damaged tissue is a testament to the accuracy of the laser ablation technique.
SE/SEM-CLEM
In SE/SEM-CLEM, cells are first imaged by light or fluorescence microscopy to capture functional or molecular information, then processed for secondary electron (SE) imaging in the SEM to visualise surface morphology. Overlaying fluorescence and SEM images enables direct correlation between cellular structure and function.
Case Study: Cortical Cytoskeleton and Cell Cycle
This approach was used in a study by Ruby Peters (Paluch Lab, Department of Physiology, Development and Neuroscience, Cambridge) to examine the actomyosin cortex—a ~150 nm thick cytoskeletal network beneath the plasma membrane that governs cell shape changes. Although mitotic and rounded interphase HeLa cells appear morphologically similar in SEM, fluorescence imaging of DNA enabled clear distinction of cell cycle phases. SEM imaging of the same cells then revealed how cortex architecture correlates with changes in cortical tension, which increases over threefold during mitosis.

Left: HeLa cells stained with DAPI - tiled overview image with marker positions taken in a Leica SP8 confocal microscope, Microscopy Bioscience Platform. Right: DAPI-SE/SEM overlays of M-phase cells at defined marker positions, scale bar is 10 μm.

After establishing the cell cycle phase using the DAPI staining and a treatment to expose the cortical cytoskeleton, the architecture of the actomyosin cortex of the same cell was analysed in more detail in SEM images at higher magnification.
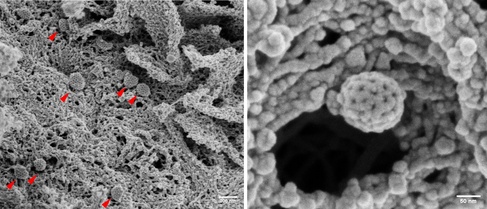
Frequently, coated pits (red arrows) were seen on the cell surface, easily recognizable by their regular clathrin network.
